 This decrease in atomic radius is due to a positive charge in the nucleus. As electrons are paired with each other, the ionization energy decreases. The outermost shell remains the same during these periods, except for the number of protons. The atomic radius of hydrogen, argon, and oxygen is measured in nanometers. The atomic radius of fluorine is smaller than that of the former. This means that the atomic size of the latter will be smaller than that of the former. When an atom has more than one electron shell, the outermost electrons are attracted to the nucleus. The atomic number and the atomic radius decrease as you go from left to right on the periodic table. For instance, helium is much smaller than hydrogen and safer to use. As you go down a group in the periodic table, atoms tend to be larger, while those on the right tend to be smaller. Moreover, their atomic sizes vary greatly. Noble gases are inert monatomic substances that are inert. The periodic table includes a group of elements called noble gases. This way, you can find any element’s atomic number and radius. The highest number of atoms is hydrogen, and the lowest number is Lithium. Second, you can find the atomic radius of a specific element by using the Periodic Table. There are two basic methods to determine atomic number and radius. 
Atomic radiusįirst, you need to know the atomic number of the element. 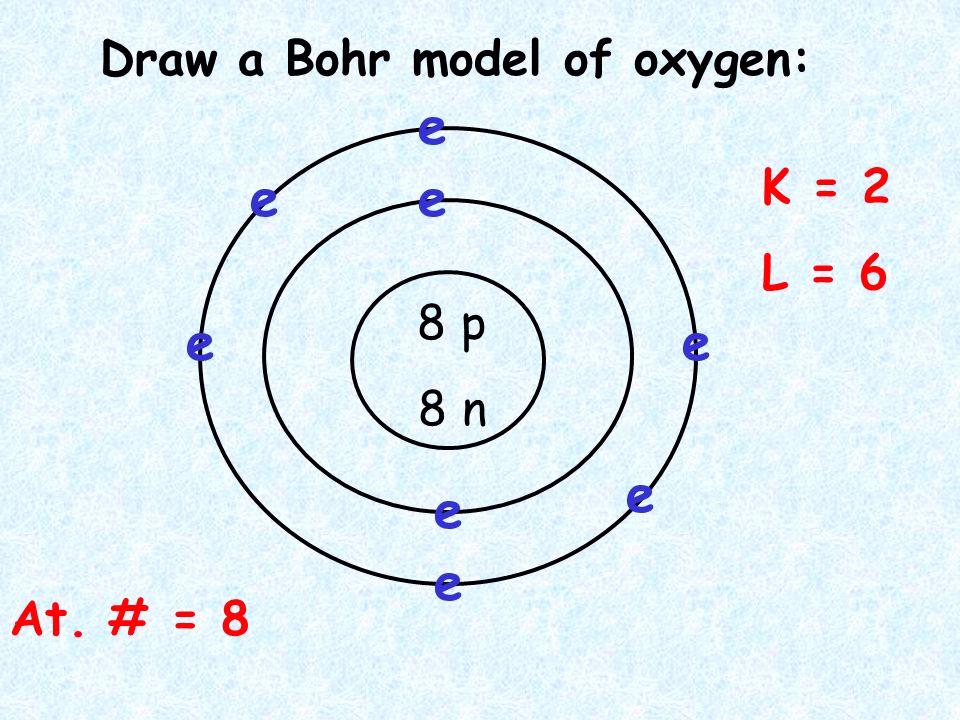
In addition, the Bohr model makes use of different electron shells that have a specific number of electrons. Each of these atoms has two electron shells, the outermost of which contains the valence electron. The Bohr model of oxygen-carbon fluorine neon li is a visual representation of electrons orbiting a small nucleus.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
